|
11/9/2023 0 Comments Zinc reactivity 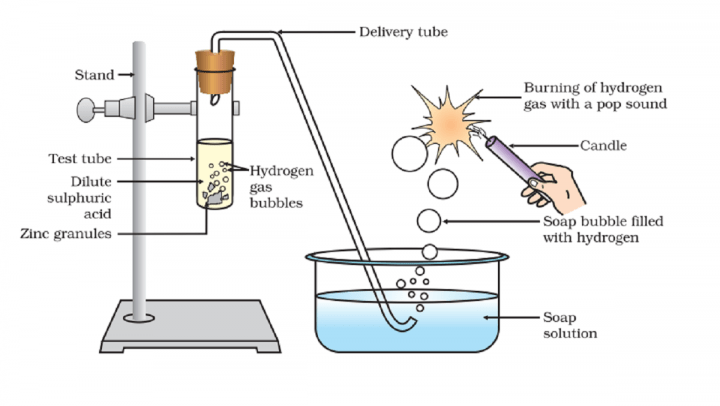
The two H free radicals combine and form H2 gas. The new unstable compound ZnCl reacts almost instantly with another HCl, forming ZnCl2 and releasing another H free radical. This happens when the Zn and HCl collide and release a H free radical. "We are always exploring new materials with properties that people thought are not attainable we do this by controlling their size, composition and charge state," he said. Actually what happens is that the Zinc displaces the H-atoms from 2 HCl molecules. Reason (R): Iron slowly reacts with oxygen and form iron oxide. Jena's groundbreaking findings on zinc build on his past work, he said, as he and colleagues have been developing atomic clusters that can be highly stable when carrying multiple charges. Q.6.Assertion (A): Rusting of iron is a slow combustion. "This is what we call modern day alchemy." 
For example, gold, a noble metal, can be reactive when size is reduced to nanometers," he said. The metal reactivity series is a commonly taught concept in chemistry, placing the metals, as its name suggests, in order of reactivity from most reactive to least reactive. "The remarkable properties of nanomaterials are that they can be very different from their bulk counterparts. Jena, the author of roughly 650 papers and 14 books, has conducted research on atomic clusters and nanoparticles for more than 35 years. Jena's paper, "Realization of Zn3+ Oxidation State," was published in the journal Nanoscale. "This study shows that fundamental chemical properties of an atom can be changed." "Its d-electrons participate in chemical reactions and zinc can carry a magnetic moment," Jena said. The order of reactivity is: magnesium > zinc > copper. However, Jena found, when reacted with highly stable trianions, zinc's properties can be changed. Use the results in the table to deduce an order of reactivity, starting with the most reactive metal. While zinc is categorized as a transition metal element, its third electron shell - arranged around the nucleus and containing electrons - is full, and unlike regular transition metals, does not take part in zinc's chemical reaction and does not allow zinc to be magnetic. "This technology allows you to manipulate chemistry at the fundamental level, making synthesis of new materials with tailored properties possible," said Jena, Distinguished Professor of Physics in the College of Humanities and Sciences. A new paper by Virginia Commonwealth University researcher Puru Jena, Ph.D., shows that the chemistry of zinc can be fundamentally changed, making it trivalent - or a valence of three - with the proper reagent. It’s also a useful tool in predicting the products of simple displacement reactions involving two different metals, as well as providing an insight into why different. Zinc traditionally has a valence of two, meaning two electrons take part in the element's chemical reaction. The metal reactivity series is a commonly taught concept in chemistry, placing the metals, as its name suggests, in order of reactivity from most reactive to least reactive. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
